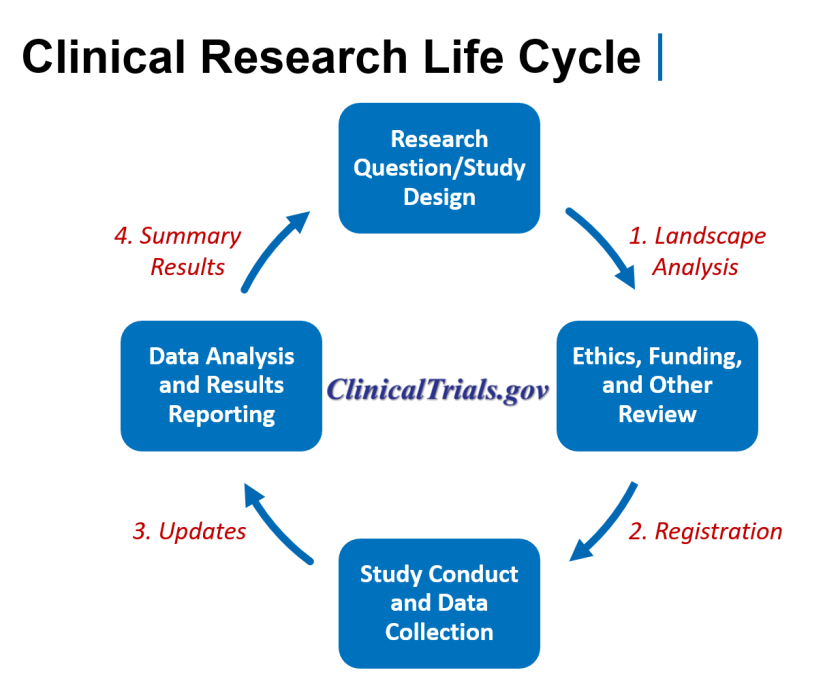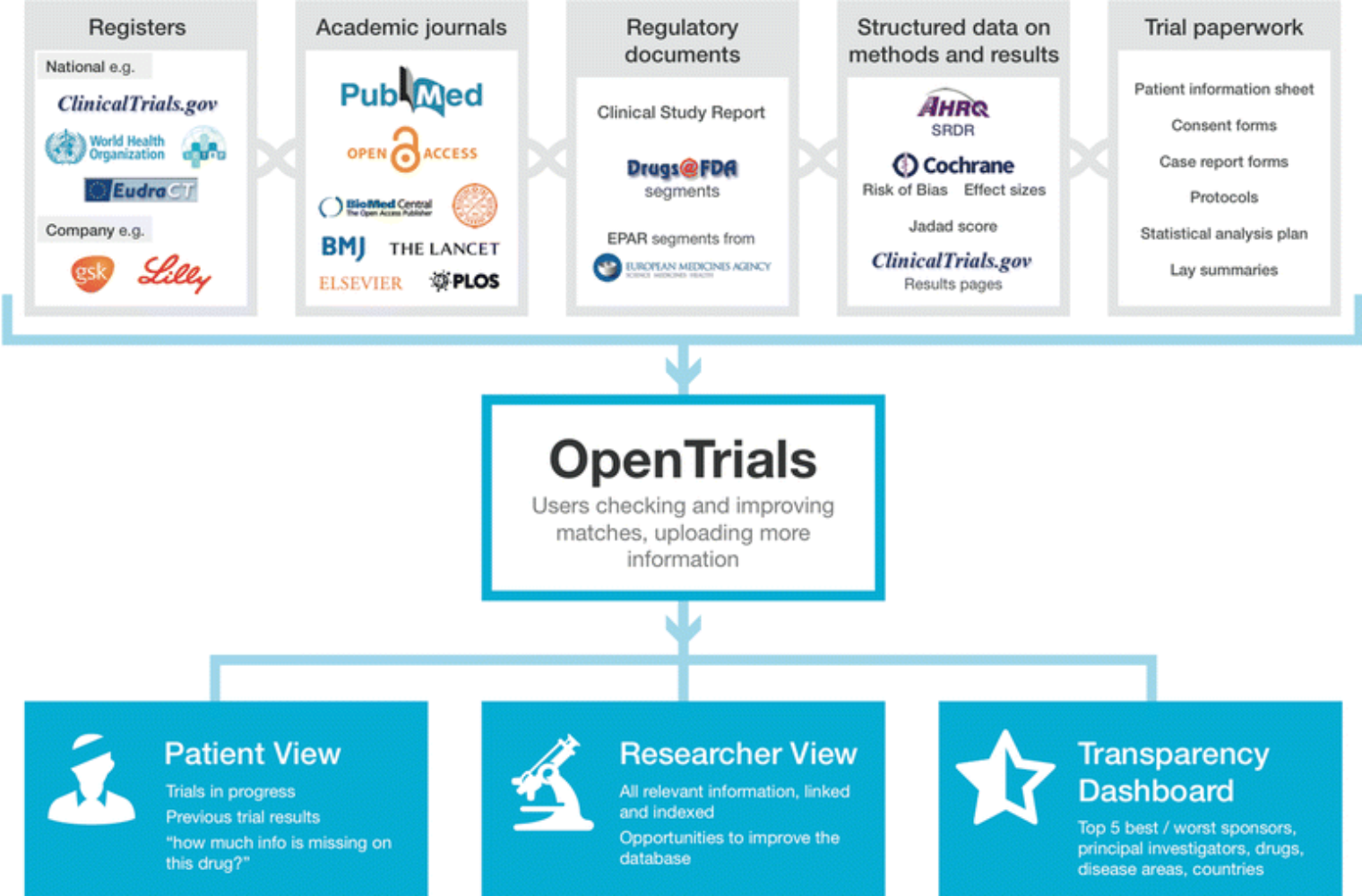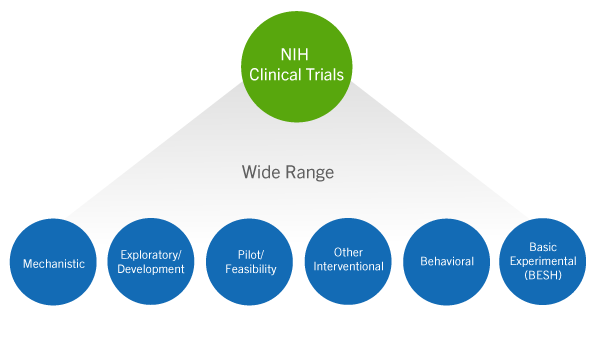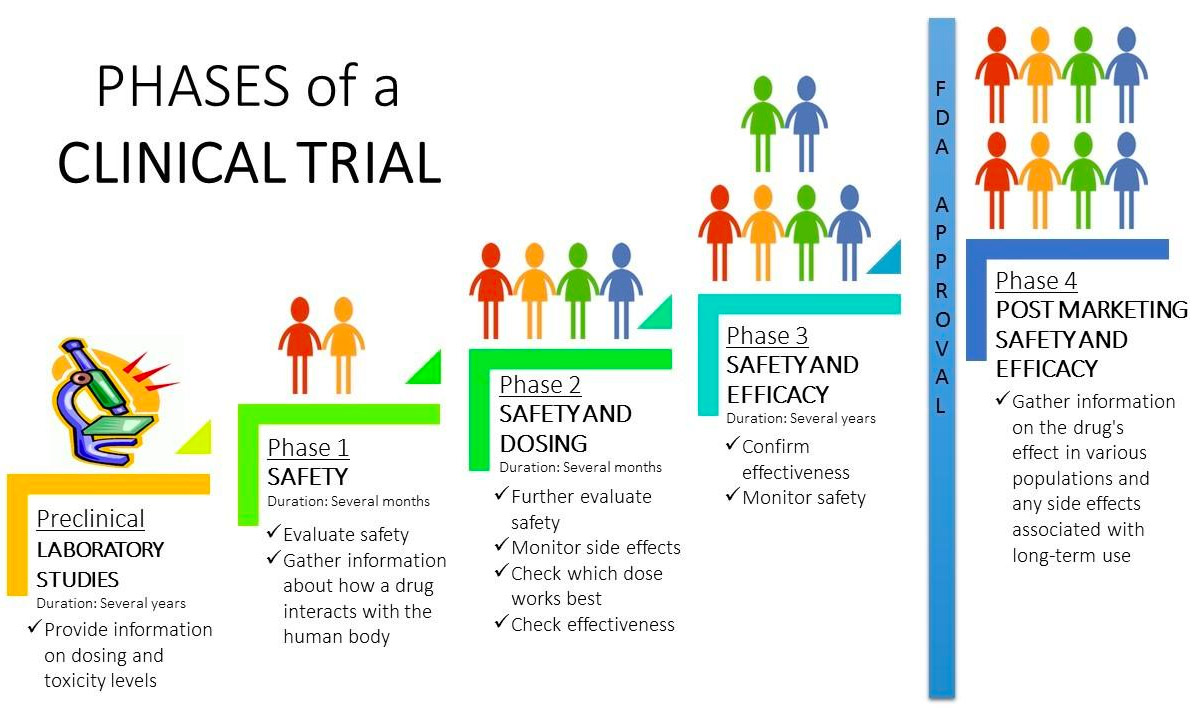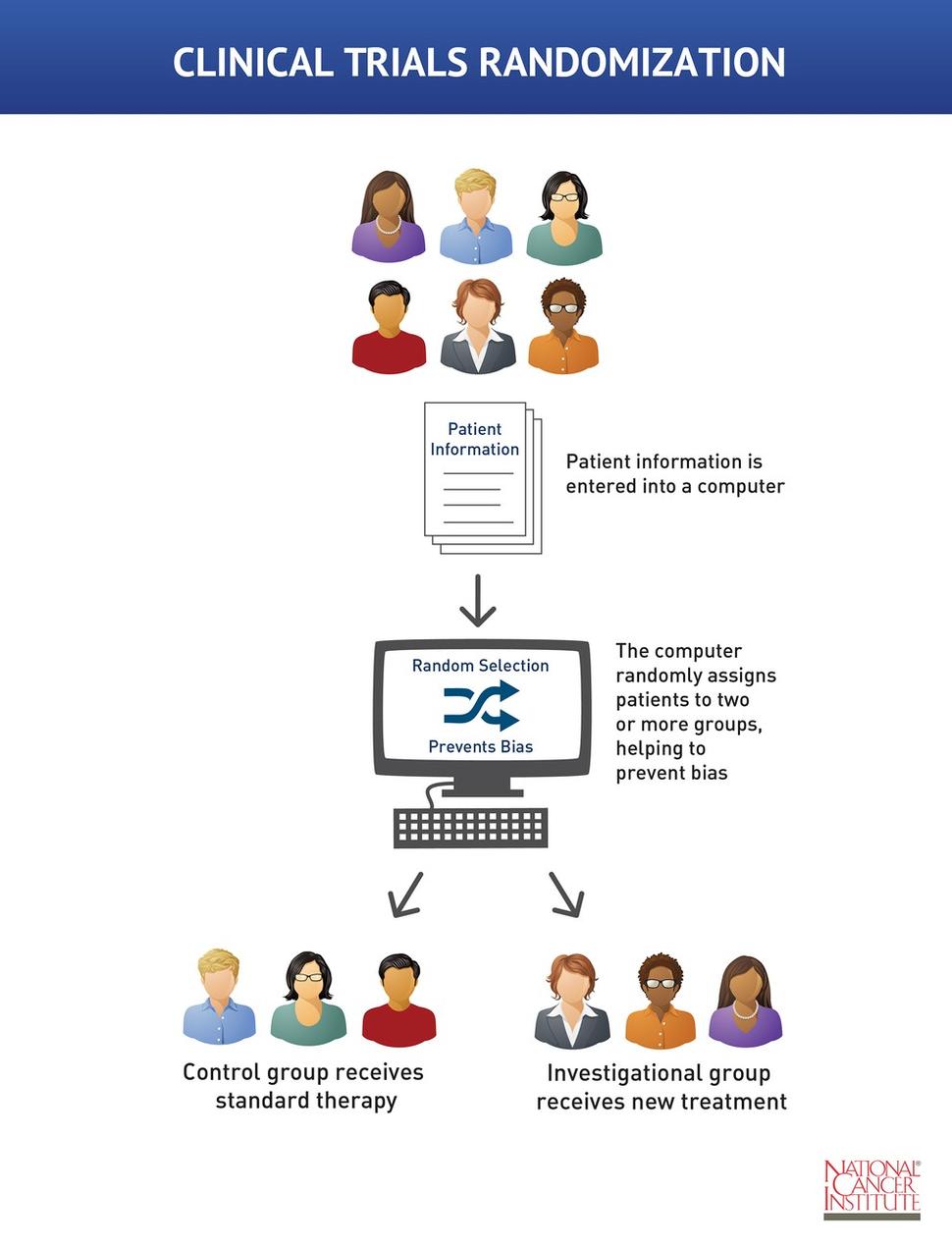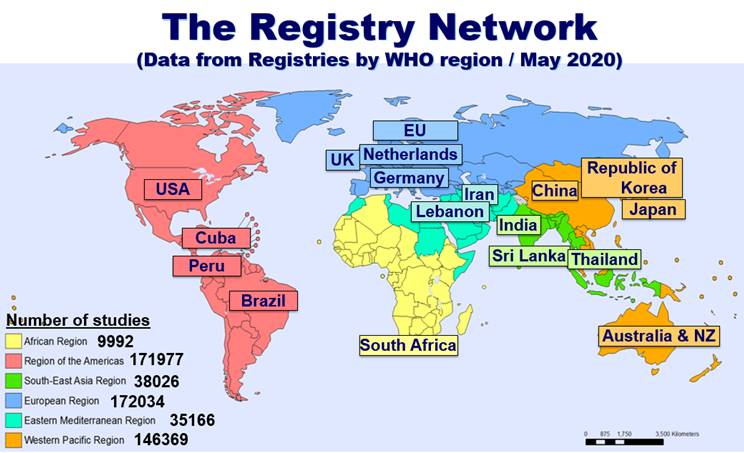
The WHO International Clinical Trials Registry Platform: Providing global clinical trial information to all - On Medicine
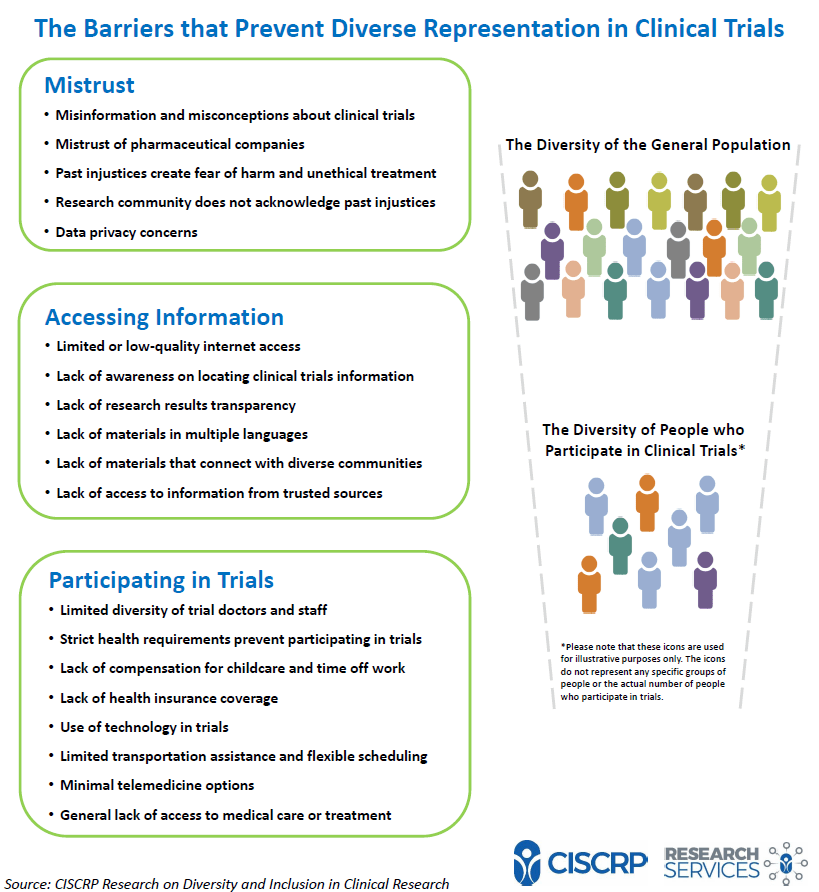
Doing Our Part: Improving Diversity in Clinical Research Participation - Center for Information & Study on Clinical Research Participation
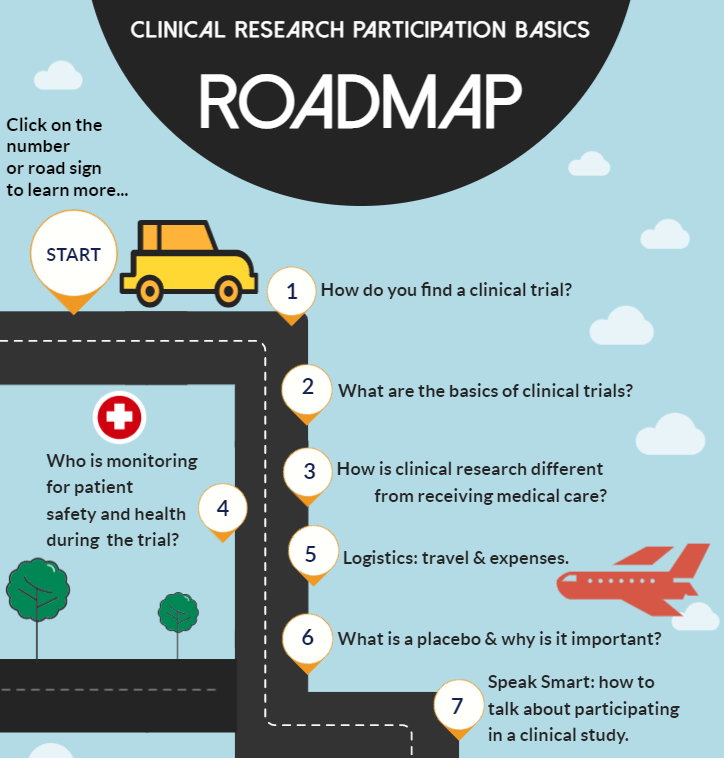
Clinical Research Participation Roadmap - Center for Information & Study on Clinical Research Participation