
Wiley Series in Probability and Statistics - 2011 - Wiley Series in Probability and Statistics - Wiley Online Library
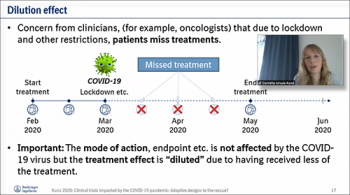
Ingram Olkin Forum Deliberates Bayes and Frequentist Approaches to Rescuing Disrupted Trials | National Institute of Statistical Sciences
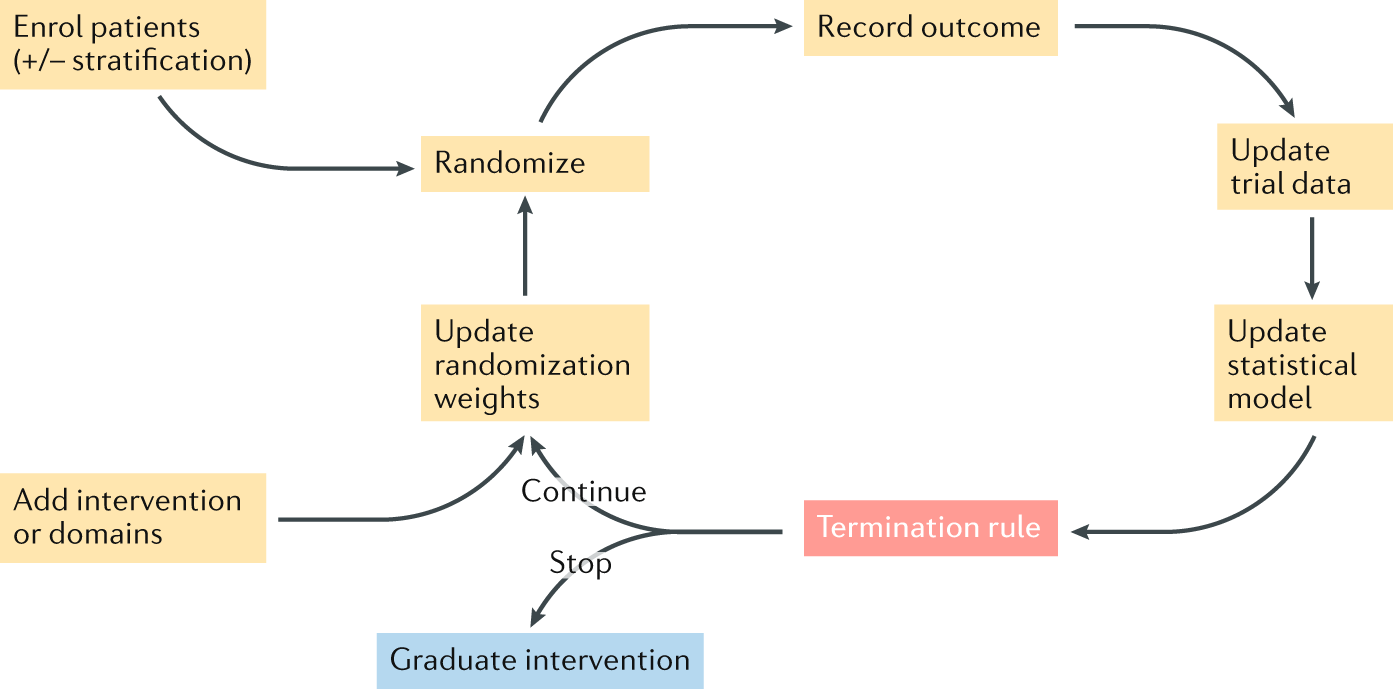
Adaptive platform trials: definition, design, conduct and reporting considerations | Nature Reviews Drug Discovery
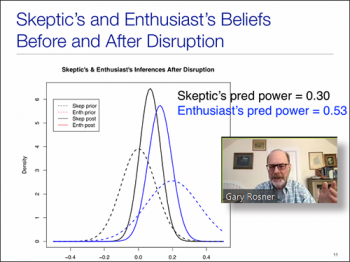
Ingram Olkin Forum Deliberates Bayes and Frequentist Approaches to Rescuing Disrupted Trials | National Institute of Statistical Sciences

Clinical trials in critical care: can a Bayesian approach enhance clinical and scientific decision making? - The Lancet Respiratory Medicine

The Evolution of Master Protocol Clinical Trial Designs: A Systematic Literature Review - Clinical Therapeutics

The Adaptive designs CONSORT Extension (ACE) statement: a checklist with explanation and elaboration guideline for reporting randomised trials that use an adaptive design | The BMJ

Bayesian statistics and clinical trial designs for human cells and tissue products for regulatory approval - ScienceDirect
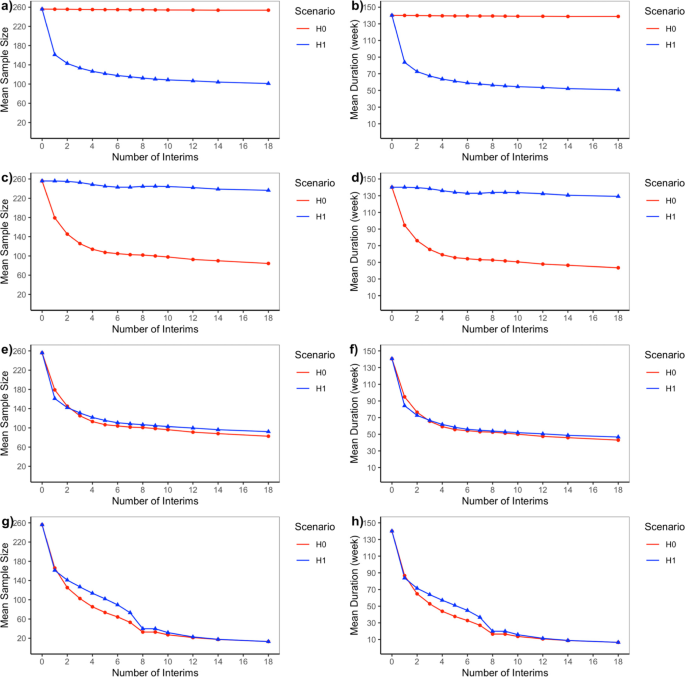
Bayesian adaptive design for pediatric clinical trials incorporating a community of prior beliefs | BMC Medical Research Methodology | Full Text
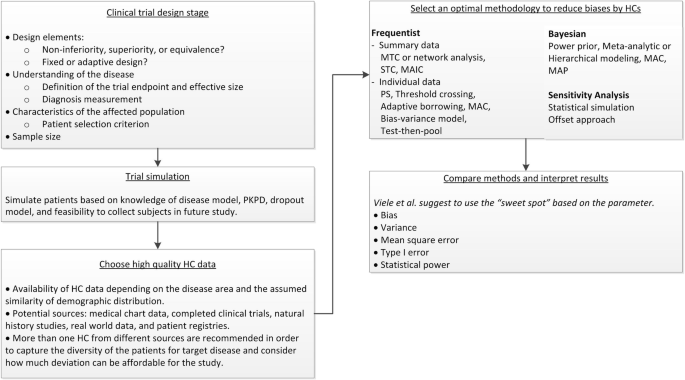




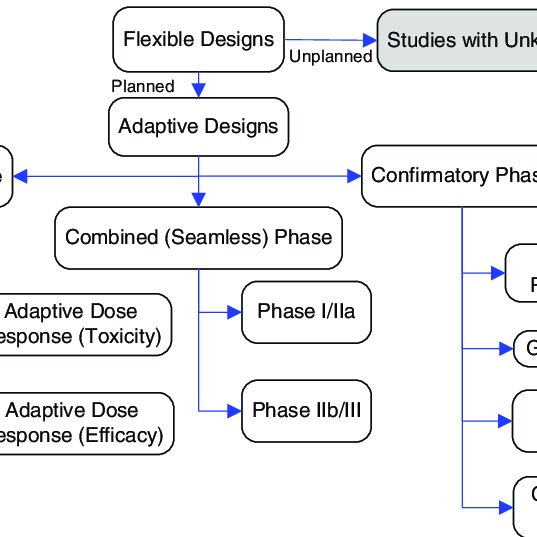


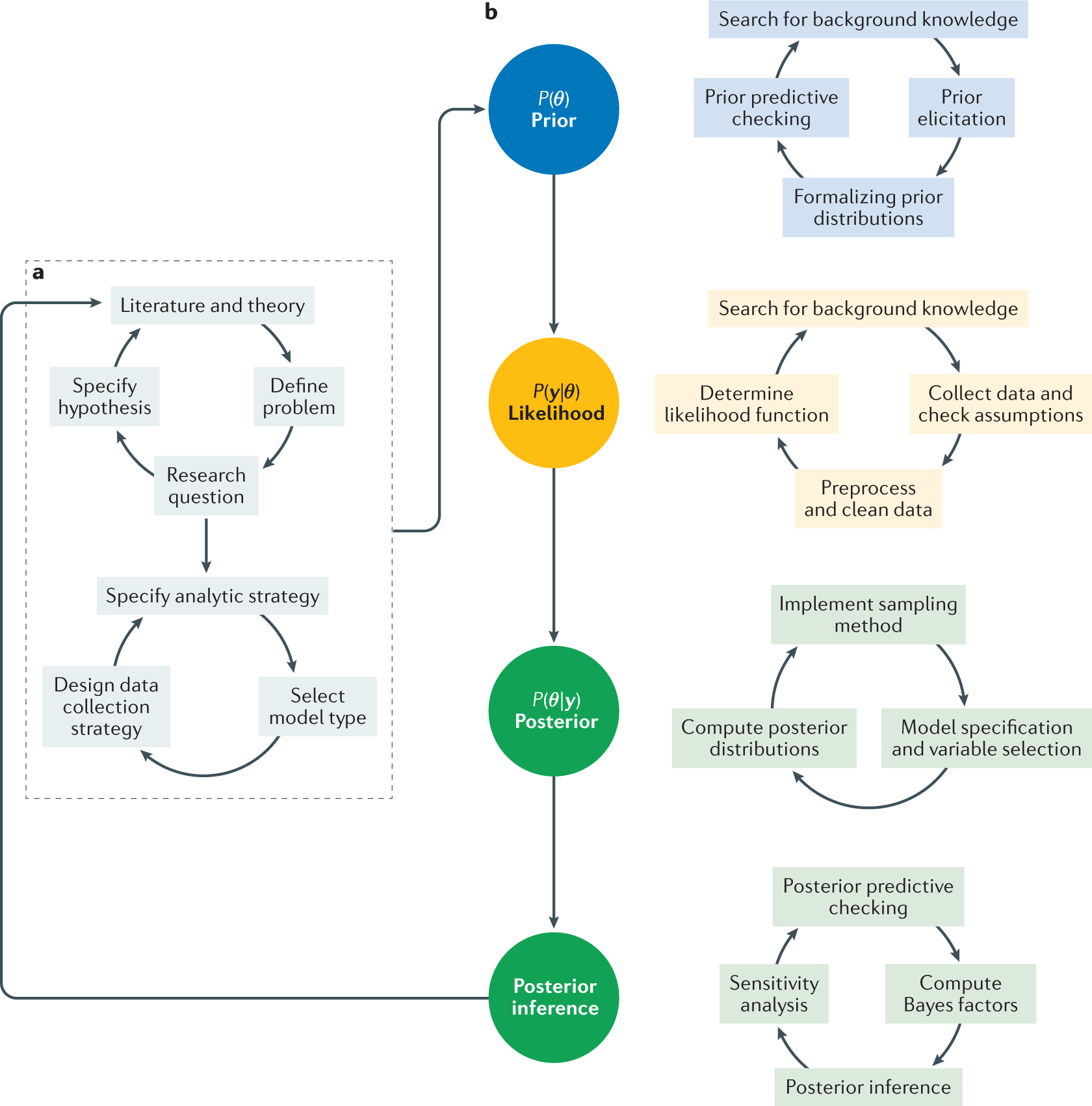

![PDF] Bayesian Adaptive Methods for Clinical Trials | Semantic Scholar PDF] Bayesian Adaptive Methods for Clinical Trials | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f71d1b9c95e95f3a11a9cb77f479f1f5ea1df605/30-Figure2-1.png)






