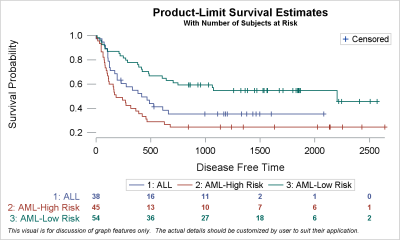
Clinical Trial Data Analysis Using R and SAS (Chapman & Hall/CRC Biostatistics Series) 2, Chen, Ding-Geng (Din), Peace, Karl E., Zhang, Pinggao - Amazon.com

Breakdown, the SAS file used by Pfzier to format it's clinical trial data for submissions to the FDA. Have fun! It's hot, just released. They wanted to wait till 2076. That seems

Clinical Trial Data Analysis Using R and SAS (Chapman & Hall/CRC Biostatistics Series): 0001498779522: Medicine & Health Science Books @ Amazon.com

Advanced Analytics is revolutionizing clinical trials: the experience of Exom Group, the first European digital CRO, to implement such an innovative approach. | Exom Group
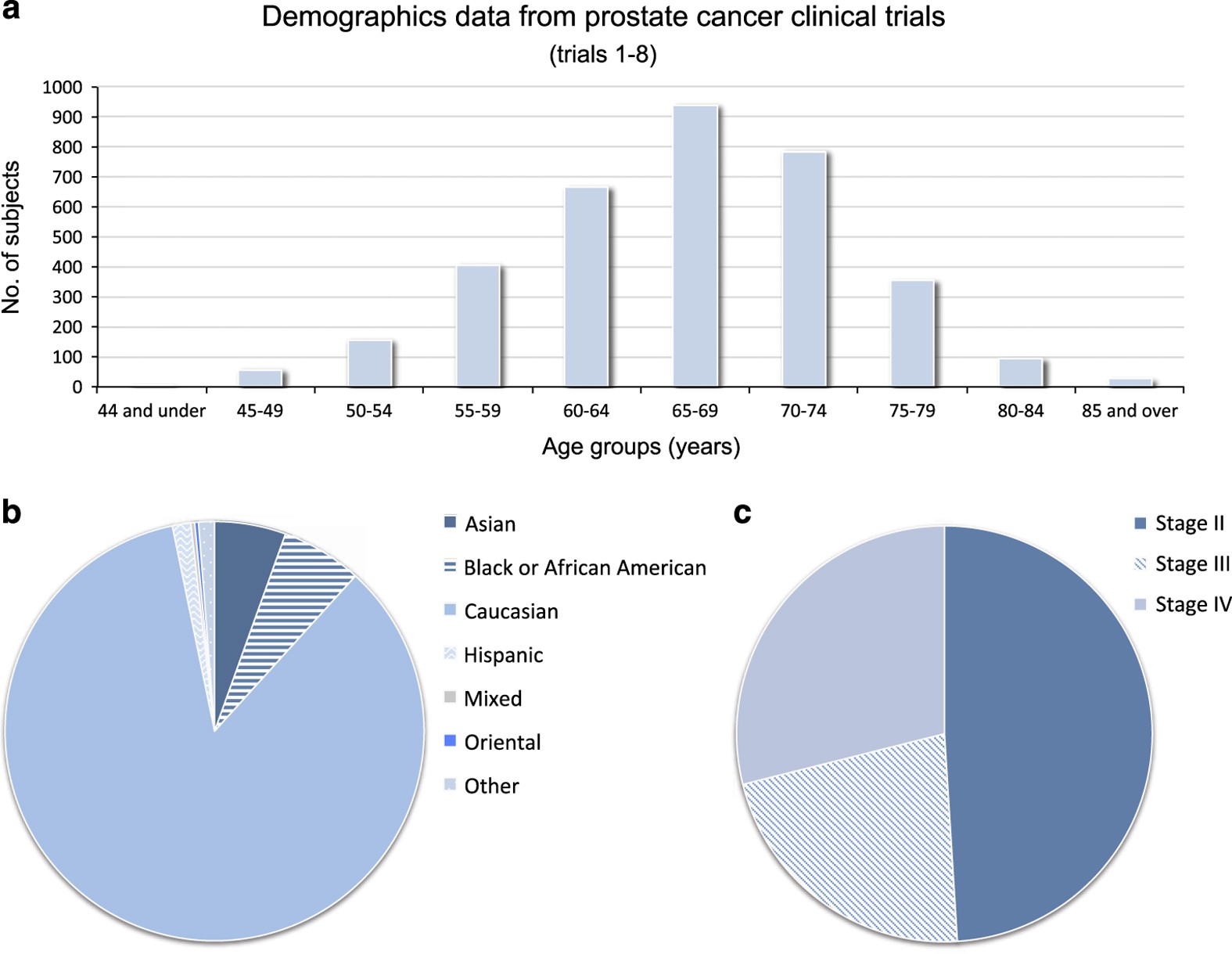
A patient-level data meta-analysis of standard-of-care treatments from eight prostate cancer clinical trials | Scientific Data